Cryptosporidiosis in Calves
Overview of the Disease
Cryptosporidiosis is caused by protozoan parasites of the genus Cryptosporidium. These are single-celled eukaryotic organisms that infect the small intestine and are primarily transmitted via the faecal–oral route.
It is a major cause of neonatal calf diarrhoea in dairy systems and a significant zoonotic pathogen, meaning it can also infect humans. Cryptosporidium parvum is the main species associated with clinical disease in pre-weaned calves in the UK (Bartley et al., 2024).
Species Diversity and Epidemiology
There are over 40 recognised species of Cryptosporidium (Prediger et al., 2021; Huang et al., 2023), but only a small number are relevant in cattle
In calves:
C. parvum is the primary pathogenic species
Other species such as C. bovis, C. ryanae, and C. andersoni are generally associated with older animals or subclinical infection
Oocyst Survival and Environmental Persistence
The infectious stage, known as the oocyst, has a thick, multi-layered wall that makes it highly resistant to environmental stress.
Oocysts:
Survive for months in moist environments (soil, water, bedding)
Are resistant to many common disinfectants
Remain infectious after excretion
This resilience makes environmental contamination a key driver of disease persistence on farms.
Zoonotic and Environmental Risk
Cryptosporidium is both an agricultural and public health concern.
Livestock density is linked to increased environmental contamination of water sources (Robinson et al., 2011)
Recreational water exposure has been associated with human cases
In 2024, cases of cryptosporidiosis were confirmed in South Devon, highlighting its ongoing zoonotic relevance
This demonstrates that cryptosporidiosis is not solely a farm-level issue but part of a wider environmental health system.
Infection Dynamics in Calves
Infection occurs via ingestion of oocysts from contaminated environments, typically:
Incubation period: 3–7 days
Peak shedding occurs in neonatal calves (5–15 days old)
Clinical signs include:
Lethargy and reduced appetite
Profuse watery diarrhoea (sometimes with mucus)
Dehydration and weight loss
A single calf can shed up to 10⁷–10⁸ oocysts per gram of faeces, leading to rapid environmental contamination and outbreak amplification in group-housed systems.
Shedding may continue even after clinical recovery, prolonging environmental contamination risk.
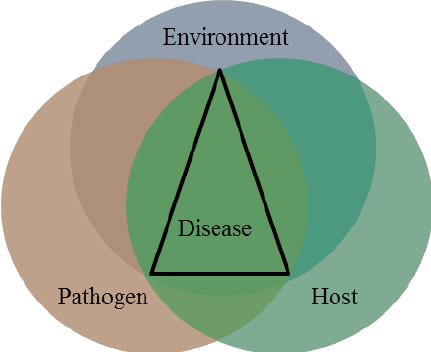
Disease Pyramid
Represents dynamics between pathogen, susceptible host and favourable environment for disease to occur.
Pathophysiology and Long-Term Effects
The parasite damages intestinal epithelial cells, leading to:
Villous atrophy
Reduced nutrient absorption
Dehydration and electrolyte imbalance
This can result in:
Poor weight gain
Reduced future productivity
Potential impacts on fertility and lifetime performance
Vaccination and Passive Immunity
A vaccine is available for use in pregnant heifers and cows in late gestation. It works by stimulating the production of anti-C. parvum antibodies, which are transferred to the calf via colostrum and transition milk.
Key points:
Protection is passive (colostrum-mediated), not direct in-calf immunity
Effectiveness depends on colostrum intake quality, timing, and hygiene
Works primarily against C. parvum, the dominant pathogenic species in calves
Studies show reduced oocyst shedding and shorter duration of diarrhoea in calves receiving antibody-rich colostrum (Mira, 2025).
Farm-Level Control Strategies (“Toolbox Approach”)
Cryptosporidiosis control relies on multiple integrated strategies:
Colostrum management
High quality, rapidly fed colostrum is essential
Avoid pooling unless hygiene is strictly controlled
Housing and stocking density
Keep calves in small, age-matched groups
Avoid mixing age classes
Hygiene and biosecurity
Clean and disinfect pens between batches
Sterilise feeding equipment (buckets, teats, tubes)
Apply strict biosecurity (footwear, clothing, handling sick calves last)
Environmental management
Maintain dry, well-bedded pens
Improve ventilation to reduce humidity
Recognise that moisture increases oocyst survival
Monitoring
Track diarrhoea patterns to identify outbreaks early
Rapid isolation and treatment of affected calves reduces spread
Summary
Cryptosporidiosis is a highly significant parasitic disease of neonatal calves caused mainly by Cryptosporidium parvum. It spreads via the faecal–oral route and is characterised by rapid environmental contamination, high oocyst shedding, and significant resilience of the infectious stage in farm environments.
The disease has important economic and zoonotic implications due to reduced calf growth, treatment costs, and potential transmission to humans through environmental contamination.
While vaccination of pregnant cows can improve passive immunity via colostrum and reduce disease severity and shedding, it does not eliminate infection. Therefore, effective control relies on a combined strategy of colostrum management, hygiene, environmental control, and stocking density reduction.
L Peddle
Reference List
Bartley, P.M., Standar, J.H. and Katzer, F. (2024). Genetic characterisation of Cryptosporidium parvum in dairy cattle and calves during the early stages of a calving season. Current research in parasitology and vector borne disease, vol 5, 100160. https://doi.org/10.1016/j.crpvbd.2023.100160
Huang, J., Chen, M., He, Y., Chen, H., Huang, M., Li, N., ... & Guo, Y. (2023). Cryptosporidium equi n. sp.(Apicomplexa: Cryptosporidiidae): biological and genetic characterisations. International journal for parasitology, 53(10), 545-554.
Lorenzo, J. M., Munekata, P. E., Dominguez, R., Pateiro, M., Saraiva, J. A., & Franco, D. (2018). Main groups of microorganisms of relevance for food safety and stability: General aspects and overall description. In Innovative technologies for food preservation (pp. 53-107). Academic Press.
Mira, A., Garro, C. J., de Alba, P., Monti, D., Lang, M. C., Vivas, A., Medina, E., Franco, J. C., Gutierrez, Á., Schnittger, L., Wigdorovitz, A., Parreño, V., & Bok, M. (2025). P23-Specific IgY Significantly Reduces Diarrhea and Oocyst Shedding in Calves Experimentally Infected with Cryptosporidium parvum. Vaccines, 13(2), 162. https://doi.org/10.3390/vaccines13020162 0029
Prediger, J., Ježková, J., Holubová, N., Sak, B., Konečný, R., Rost, M., McEvoy, J., Rajský, D., & Kváč, M. (2021). Cryptosporidium sciurinum n. sp. (Apicomplexa: Cryptosporidiidae) in Eurasian Red Squirrels (Sciurus vulgaris). Microorganisms, 9(10), 2050. https://doi.org/10.3390/microorganisms9102050
Robinson, G., Chalmers, R. M., Stapleton, C., Palmer, S. R., Watkins, J., Francis, C., & Kay, D. (2011). ). A whole water catchment approach to investigating the origin and distribution of Cryptosporidium species. Journal of Applied Microbiology, 111(3), 717-730.)
Shaw, H. J., Armstrong, C., Uttley, K., Morrison, L. J., Innes, E. A., & Katzer, F. (2021). Genetic diversity and shedding profiles for Cryptosporidium parvum in adult cattle and their calves. Current Research in Parasitology & Vector-Borne Diseases, 1, 100027.